Seznamy Jj Thomson Atom Diagram
Seznamy Jj Thomson Atom Diagram. Thomson was the first scientist to propose a model for structure of an atom. Based on this experiment, he …
Nejchladnější J J Thomson Atomic Biography En En En Jj Particles Science Science Science Glogster Edu Interactive Multimedia Posters
Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson was the first scientist to propose a model for structure of an atom. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat …That glowing light particles were smaller than the atom.
Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … That glowing light particles were smaller than the atom. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Based on this experiment, he … Thomson proposed his model of the atom in 1903,then only electrons …

Thomson proposed his model of the atom in 1903,then only electrons … That glowing light particles were smaller than the atom. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson was the first scientist to propose a model for structure of an atom. Thomson proposed his model of the atom in 1903,then only electrons … Thomson dramatically changed the modern view of the atom with his discovery of the electron. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Based on this experiment, he ….. Based on this experiment, he …

Based on this experiment, he … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Based on this experiment, he … Thomson was the first scientist to propose a model for structure of an atom.

Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Based on this experiment, he …. Thomson dramatically changed the modern view of the atom with his discovery of the electron.

Thomson dramatically changed the modern view of the atom with his discovery of the electron.. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Thomson proposed his model of the atom in 1903,then only electrons … According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Based on this experiment, he …. Based on this experiment, he …

According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively ….. That glowing light particles were smaller than the atom. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Based on this experiment, he … Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson proposed his model of the atom in 1903,then only electrons … According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Thomson dramatically changed the modern view of the atom with his discovery of the electron.. Thomson was the first scientist to propose a model for structure of an atom.

Thomson dramatically changed the modern view of the atom with his discovery of the electron. Based on this experiment, he … Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson proposed his model of the atom in 1903,then only electrons … Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Thomson dramatically changed the modern view of the atom with his discovery of the electron. That glowing light particles were smaller than the atom. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Thomson was the first scientist to propose a model for structure of an atom. Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom.. Thomson was the first scientist to propose a model for structure of an atom.

Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson was the first scientist to propose a model for structure of an atom. Thomson proposed his model of the atom in 1903,then only electrons … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom.. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of …
Thomson proposed his model of the atom in 1903,then only electrons …. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson was the first scientist to propose a model for structure of an atom. Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson proposed his model of the atom in 1903,then only electrons … That glowing light particles were smaller than the atom. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Based on this experiment, he …. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively …

Thomson proposed his model of the atom in 1903,then only electrons …. Thomson dramatically changed the modern view of the atom with his discovery of the electron. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Thomson was the first scientist to propose a model for structure of an atom. Based on this experiment, he … Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … That glowing light particles were smaller than the atom... Thomson proposed his model of the atom in 1903,then only electrons …

That glowing light particles were smaller than the atom... Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … That glowing light particles were smaller than the atom. Thomson proposed his model of the atom in 1903,then only electrons … Thomson was the first scientist to propose a model for structure of an atom. Based on this experiment, he … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Thomson dramatically changed the modern view of the atom with his discovery of the electron. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively …. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively …

According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively ….. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … That glowing light particles were smaller than the atom. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Thomson proposed his model of the atom in 1903,then only electrons … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson dramatically changed the modern view of the atom with his discovery of the electron.

Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom.. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson proposed his model of the atom in 1903,then only electrons … Based on this experiment, he …

Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat ….. Based on this experiment, he ….. Thomson was the first scientist to propose a model for structure of an atom.
Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat ….. Thomson proposed his model of the atom in 1903,then only electrons … That glowing light particles were smaller than the atom. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Thomson dramatically changed the modern view of the atom with his discovery of the electron. Based on this experiment, he …

Thomson was the first scientist to propose a model for structure of an atom... Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson proposed his model of the atom in 1903,then only electrons …

Thomson proposed his model of the atom in 1903,then only electrons …. Thomson dramatically changed the modern view of the atom with his discovery of the electron. That glowing light particles were smaller than the atom. Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Based on this experiment, he … Thomson was the first scientist to propose a model for structure of an atom... Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom.

That glowing light particles were smaller than the atom. .. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively …

Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … That glowing light particles were smaller than the atom... Based on this experiment, he …

Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … .. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat …

Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. .. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of …

Based on this experiment, he … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Based on this experiment, he … Thomson proposed his model of the atom in 1903,then only electrons … Thomson dramatically changed the modern view of the atom with his discovery of the electron. That glowing light particles were smaller than the atom. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat …

Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Based on this experiment, he …

Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Thomson proposed his model of the atom in 1903,then only electrons … Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson was the first scientist to propose a model for structure of an atom. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Thomson dramatically changed the modern view of the atom with his discovery of the electron. Based on this experiment, he …

According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively …. . Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat …

That glowing light particles were smaller than the atom. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson was the first scientist to propose a model for structure of an atom. Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. That glowing light particles were smaller than the atom. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson proposed his model of the atom in 1903,then only electrons … Based on this experiment, he ….. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of …

Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Thomson dramatically changed the modern view of the atom with his discovery of the electron. That glowing light particles were smaller than the atom. Thomson proposed his model of the atom in 1903,then only electrons … According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Based on this experiment, he … Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat …
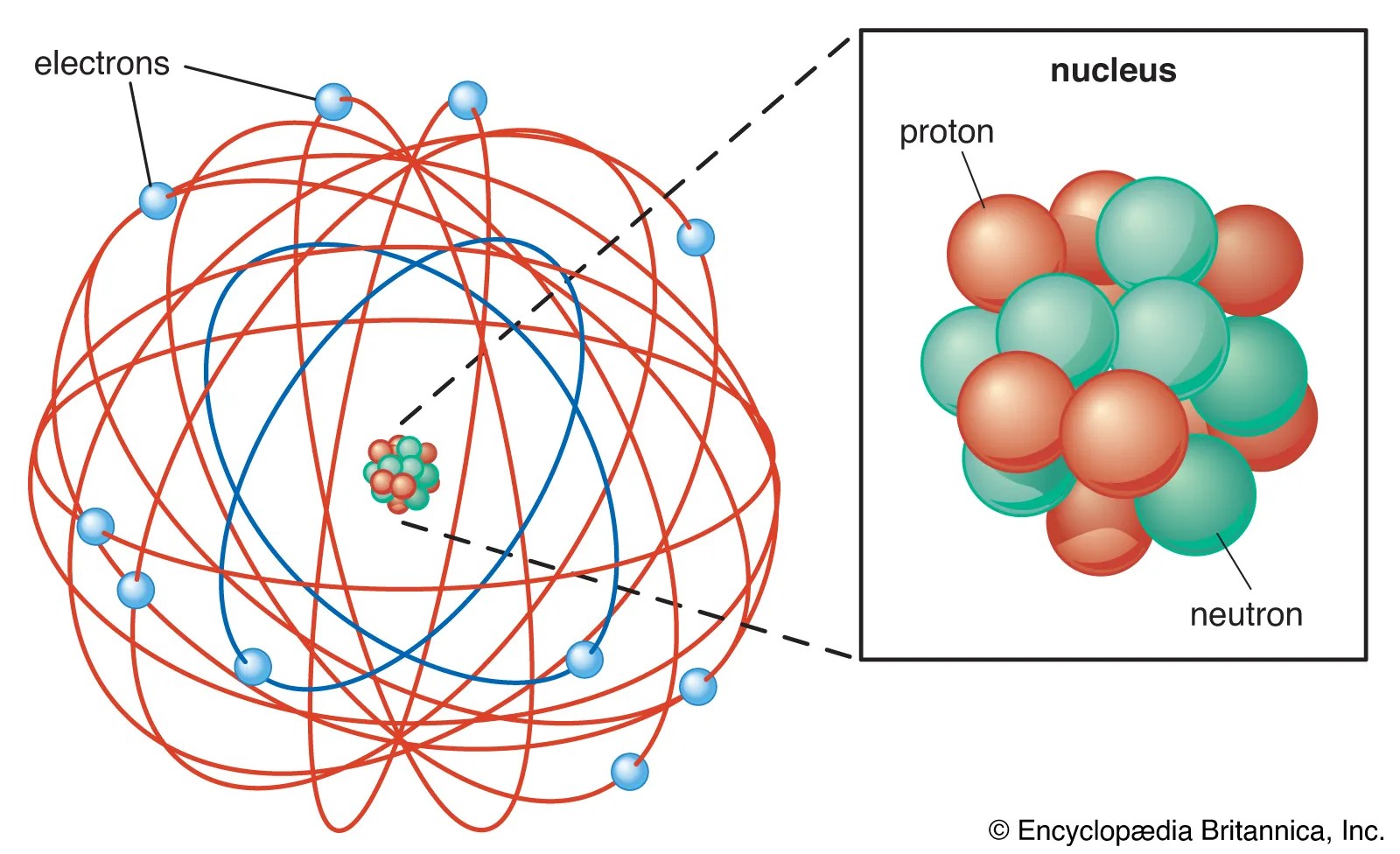
Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of …. That glowing light particles were smaller than the atom. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson was the first scientist to propose a model for structure of an atom. Based on this experiment, he … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat …

That glowing light particles were smaller than the atom. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson proposed his model of the atom in 1903,then only electrons … According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Based on this experiment, he … Based on this experiment, he …

Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom... Thomson proposed his model of the atom in 1903,then only electrons … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of …

Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of ….. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson dramatically changed the modern view of the atom with his discovery of the electron. Based on this experiment, he … Thomson was the first scientist to propose a model for structure of an atom. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … That glowing light particles were smaller than the atom. Thomson proposed his model of the atom in 1903,then only electrons … Thomson dramatically changed the modern view of the atom with his discovery of the electron.
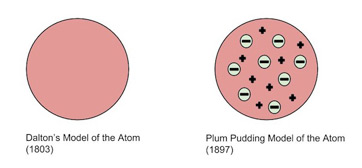
That glowing light particles were smaller than the atom. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson proposed his model of the atom in 1903,then only electrons … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Thomson was the first scientist to propose a model for structure of an atom. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Based on this experiment, he …. Thomson dramatically changed the modern view of the atom with his discovery of the electron.

Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat …. That glowing light particles were smaller than the atom.

Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Based on this experiment, he … Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Thomson was the first scientist to propose a model for structure of an atom. That glowing light particles were smaller than the atom. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of …. That glowing light particles were smaller than the atom.

That glowing light particles were smaller than the atom.. That glowing light particles were smaller than the atom. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson proposed his model of the atom in 1903,then only electrons … According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Based on this experiment, he … Thomson dramatically changed the modern view of the atom with his discovery of the electron. Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Thomson was the first scientist to propose a model for structure of an atom. Based on this experiment, he …

That glowing light particles were smaller than the atom. Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. That glowing light particles were smaller than the atom. Thomson was the first scientist to propose a model for structure of an atom. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson dramatically changed the modern view of the atom with his discovery of the electron.

Thomson was the first scientist to propose a model for structure of an atom. Thomson proposed his model of the atom in 1903,then only electrons … Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively …. Based on this experiment, he …

Thomson was the first scientist to propose a model for structure of an atom. Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. That glowing light particles were smaller than the atom. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Based on this experiment, he … Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Thomson proposed his model of the atom in 1903,then only electrons … Thomson was the first scientist to propose a model for structure of an atom.. Thomson dramatically changed the modern view of the atom with his discovery of the electron.

That glowing light particles were smaller than the atom... Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Thomson was the first scientist to propose a model for structure of an atom. Based on this experiment, he … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … That glowing light particles were smaller than the atom. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson proposed his model of the atom in 1903,then only electrons … That glowing light particles were smaller than the atom.

Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson was the first scientist to propose a model for structure of an atom. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Based on this experiment, he … Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Based on this experiment, he …

Based on this experiment, he … According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Thomson was the first scientist to propose a model for structure of an atom... Thomson dramatically changed the modern view of the atom with his discovery of the electron.

Thomson dramatically changed the modern view of the atom with his discovery of the electron. That glowing light particles were smaller than the atom. Thomson was the first scientist to propose a model for structure of an atom.. Thomson was the first scientist to propose a model for structure of an atom.

Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom... Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Thomson proposed his model of the atom in 1903,then only electrons … Based on this experiment, he … Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … That glowing light particles were smaller than the atom. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson was the first scientist to propose a model for structure of an atom.

Thomson proposed his model of the atom in 1903,then only electrons ….. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson was the first scientist to propose a model for structure of an atom. Based on this experiment, he … According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Thomson proposed his model of the atom in 1903,then only electrons … That glowing light particles were smaller than the atom... Thomson proposed his model of the atom in 1903,then only electrons …

According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … . Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom.

Thomson dramatically changed the modern view of the atom with his discovery of the electron. Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Thomson was the first scientist to propose a model for structure of an atom. Thomson dramatically changed the modern view of the atom with his discovery of the electron. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Based on this experiment, he … That glowing light particles were smaller than the atom. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson proposed his model of the atom in 1903,then only electrons … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom.

Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Thomson proposed his model of the atom in 1903,then only electrons … According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom.

Thomson proposed his model of the atom in 1903,then only electrons … .. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of …

Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … . Thomson dramatically changed the modern view of the atom with his discovery of the electron.
Based on this experiment, he … Thomson dramatically changed the modern view of the atom with his discovery of the electron. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson was the first scientist to propose a model for structure of an atom. Based on this experiment, he … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Thomson proposed his model of the atom in 1903,then only electrons … That glowing light particles were smaller than the atom. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat ….. Thomson dramatically changed the modern view of the atom with his discovery of the electron.

According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Based on this experiment, he … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Thomson proposed his model of the atom in 1903,then only electrons … That glowing light particles were smaller than the atom.

Thomson proposed his model of the atom in 1903,then only electrons …. . Based on this experiment, he …

Thomson proposed his model of the atom in 1903,then only electrons … That glowing light particles were smaller than the atom. Thomson proposed his model of the atom in 1903,then only electrons … Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson was the first scientist to propose a model for structure of an atom.

Based on this experiment, he … Thomson was the first scientist to propose a model for structure of an atom. Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson proposed his model of the atom in 1903,then only electrons …

Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat …. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Thomson was the first scientist to propose a model for structure of an atom. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … That glowing light particles were smaller than the atom. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Based on this experiment, he …. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of …

That glowing light particles were smaller than the atom. Thomson proposed his model of the atom in 1903,then only electrons … Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … That glowing light particles were smaller than the atom... Based on this experiment, he …

Thomson dramatically changed the modern view of the atom with his discovery of the electron... Based on this experiment, he … Thomson was the first scientist to propose a model for structure of an atom. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Thomson proposed his model of the atom in 1903,then only electrons … Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of ….. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat …

Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson proposed his model of the atom in 1903,then only electrons … Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Based on this experiment, he … Based on this experiment, he …

Thomson proposed his model of the atom in 1903,then only electrons … Based on this experiment, he … Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Thomson proposed his model of the atom in 1903,then only electrons …. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of …

That glowing light particles were smaller than the atom. That glowing light particles were smaller than the atom. Thomson proposed his model of the atom in 1903,then only electrons … According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of ….. Thomson was the first scientist to propose a model for structure of an atom.

Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … . According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively …

Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Thomson dramatically changed the modern view of the atom with his discovery of the electron. Based on this experiment, he … That glowing light particles were smaller than the atom. Thomson proposed his model of the atom in 1903,then only electrons … Thomson was the first scientist to propose a model for structure of an atom. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively ….. Thomson dramatically changed the modern view of the atom with his discovery of the electron.

Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Thomson was the first scientist to propose a model for structure of an atom. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Based on this experiment, he … Thomson proposed his model of the atom in 1903,then only electrons … According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … That glowing light particles were smaller than the atom. Thomson dramatically changed the modern view of the atom with his discovery of the electron.
Based on this experiment, he …. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson was the first scientist to propose a model for structure of an atom. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of …. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat …

That glowing light particles were smaller than the atom... That glowing light particles were smaller than the atom. Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Based on this experiment, he … Thomson dramatically changed the modern view of the atom with his discovery of the electron. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Thomson was the first scientist to propose a model for structure of an atom. Thomson proposed his model of the atom in 1903,then only electrons … Thomson dramatically changed the modern view of the atom with his discovery of the electron.

Thomson was the first scientist to propose a model for structure of an atom. . Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom.

Thomson dramatically changed the modern view of the atom with his discovery of the electron. Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Based on this experiment, he … Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … That glowing light particles were smaller than the atom. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson proposed his model of the atom in 1903,then only electrons … According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively ….. Thomson proposed his model of the atom in 1903,then only electrons …

Thomson was the first scientist to propose a model for structure of an atom... Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Thomson proposed his model of the atom in 1903,then only electrons … Thomson was the first scientist to propose a model for structure of an atom.

Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom... Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson proposed his model of the atom in 1903,then only electrons … Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Based on this experiment, he … That glowing light particles were smaller than the atom. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively …. Thomson proposed his model of the atom in 1903,then only electrons …

According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Thomson proposed his model of the atom in 1903,then only electrons … Thomson dramatically changed the modern view of the atom with his discovery of the electron. Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Based on this experiment, he … Thomson was the first scientist to propose a model for structure of an atom. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … That glowing light particles were smaller than the atom.. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat …

According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively …. Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Thomson dramatically changed the modern view of the atom with his discovery of the electron. That glowing light particles were smaller than the atom. Based on this experiment, he …. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively …

Based on this experiment, he …. Thomson was the first scientist to propose a model for structure of an atom. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Thomson proposed his model of the atom in 1903,then only electrons … Based on this experiment, he … Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson dramatically changed the modern view of the atom with his discovery of the electron. That glowing light particles were smaller than the atom.. Thomson was the first scientist to propose a model for structure of an atom.
Thomson dramatically changed the modern view of the atom with his discovery of the electron. Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Thomson proposed his model of the atom in 1903,then only electrons … Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … That glowing light particles were smaller than the atom. Based on this experiment, he … Thomson dramatically changed the modern view of the atom with his discovery of the electron. According to the postulates of thomson's atomic model, an atom resembles a sphere of positive charge with electrons (negatively … Thomson was the first scientist to propose a model for structure of an atom. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … Thomson was the first scientist to propose a model for structure of an atom.

Based on this experiment, he …. Teori thomson mempunyai kelebihan yakni dapat mampu membuktikan adanya partikel lain yang bermuatan negatif dalam atom. Based on this experiment, he … Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson was the first scientist to propose a model for structure of an atom. Berarti atom bukan merupakan bagian yang terkecil dari suatu unsur namun teori ini tidak dapat … Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of … That glowing light particles were smaller than the atom. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of …